A drug for acute NAION is currently in phase III clinical trials
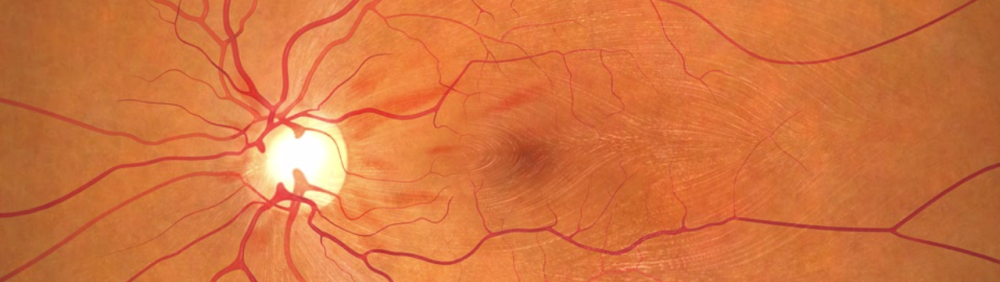
After glaucoma, the second-most common source of optic nerve damage is Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION). We usually describe this to a patient as a “stroke in the eye,” where blood supply to the optic nerve is cut off due to conditions ranging from hypotension to carotid occlusive disease. This condition typically causes sudden and acute vision loss in one eye, without any pain. Some patients get marginal improvement with time, but usually the prognosis is poor.
Given how common this blinding condition is, it’s shocking that there has been no effective treatment for this condition—until now. A new drug is currently in phase III clinical trials.
Many NAION treatments attempted, but ineffective
Many treatments for NAION have been attempted, but corticosteroids including high concentration IV steroids and normobaric oxygen therapy have not been conclusively effective in clinical trials.
A drug for acute NAION has reached phase III clinical trials.
Quark Pharmaceuticals and NORDIC are collaborating on an investigational drug trial to evaluate a neuroprotection therapy for acute NAION. The drug (QPI-1007) is not yet FDA approved. The drug is currently in phase III clinical trials. Continuous monitoring by an independent data monitoring committee (DMC) confirms the drug’s safety. Clinical trials for such patients are difficult to fill because doctors are not often aware of pipeline drug treatments and study site locations.
Details of the clinical trial
This drug study is a randomized, double-masked, sham-controlled trial delivered by a single or multi-dose intravitreal injection(s) to subjects with NAION. It will involve studying the effects on visual function in subjects with recent-onset NAION, as well as safety and tolerability in addition to structural changes in the retina. Subjects are randomized into one of three groups with only one group being a sham treatment. The three cohorts include a low dose and a high dose of either a single or multiple injections, and a sham arm.
To qualify for the study, patients must be between age 50-80 with their first episode of NAION within the last 14 days.
To qualify for the study patients must be between ages 50 and 80 with a positive diagnosis of a first episode of NAION within the last 14 days. The trial is being conducted by neuro-ophthalmologists and retina specialists in private practices and in hospitals around the US and in 6 other countries (India, China, Israel, Germany, Italy, and Australia).
The importance of visual patient education
It’s essential for ophthalmologists to educate their patients on the optic nerve and the retina. Visual tools can help illustrate conditions like NAION to patients, help them understand the inner workings of the eye’s anatomy, and come to you when they experience any change or problem with their vision. Narrated animations can complement your explanations of conditions like NAION. Rendia’s Exam Mode can help illustrate these concepts:
It’s always gratifying to write about treatments for previously untreatable blinding conditions, with hopes that more patients’ vision may be saved in the future.
Mark your calendar! Join Rendia and Dr. Paul Karpecki in discussing new approaches to #Myopia Management on October 19th, 2021 at 12:00 PM EDT! Register here!