Increasing awareness among glaucoma patients undergoing cataract surgery
There are about 3 million people over the age of 40 diagnosed with glaucoma in the U.S. It is one of the top two leading causes of blindness in this patient population, and the number is expected to triple by 2050. Yet only about 50 percent of patients know they have this blinding disease—and more than half of patients with glaucoma who undergo cataract surgery miss out on the opportunity to have a simultaneous surgery that could allow them to avoid long-term glaucoma therapy. Are you and your patients aware of the latest innovation in MIGS?
MIGS: a better solution for glaucoma patients
Minimally invasive glaucoma surgery, or MIGS, is a significant innovation for the management of glaucoma. Although topical drops have served the majority of patients in the U.S., surgical procedures like selective laser trabeculoplasty (SLT) and now MIGS comprise a larger percentage of treatment options. MIGS is best indicated for the mild to moderate glaucoma patient.
While drops are the main treatment prescribed to glaucoma patients, patient adherence is an issue, as is the potential development of ocular surface disease (OSD). In fact, only 10 percent of glaucoma patients continuously refill their prescriptions over 12 months. Patients who have been on glaucoma therapy are more than five times more likely to develop OSD, and patients who develop OSD are less compliant with medications. Studies show that there is more than three times the risk of progression of glaucoma in patients with OSD.
Drops are the usual treatment prescribed to glaucoma patients, but adherence and ocular surface disease are issues. MIGS is a better solution, since it often eliminates the need for drops.
MIGS is a better solution. It often eliminates the need for glaucoma drops. According to data from Ivantis’s FDA pivotal trial for the Hydrus Microstent, 72 percent of patients on one medication preoperatively did not require any medication through four years following MIGS. In addition, MIGS is covered by Medicare and most other insurance providers, so there is no cost to the patient.
In the U.S., MIGS typically requires performing the procedure at the time of cataract surgery. Of the 3.7 million cataract surgeries performed per year, approximately 20 percent of those patients have primary open-angle glaucoma—meaning around 800,000 patients could be eligible for a MIGS procedure. Unfortunately, the number is significantly lower.
Why are so many glaucoma patients missing out on this ideal solution, if it’s not due to cost or efficacy? The reason is lack of awareness.
The Hydrus four-year data results
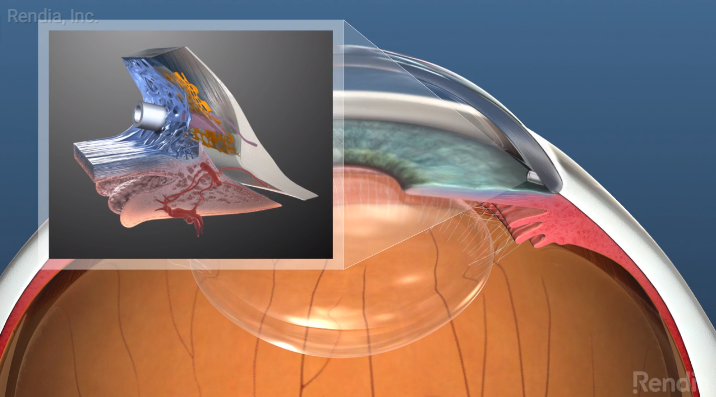
MIGS has come a long way since the first microstents were developed more than 10 years ago. They were often difficult to place correctly and did not significantly reduce pressures. The Hydrus microstent, approved by the FDA in 2018, is the only MIGS procedure that is canal restoring and covers 90 degrees of Schlemm’s canal. It’s a flexible 8mm stent composed of nitinol, which is the same material that makes up the vast majority of heart stents. It’s contoured to match the canal curvature and has three open windows facing the anterior chamber.
Nearly 70 percent of patients treated with Hydrus implants were still medication-free four years later.
In the FDA pivotal trial, nearly 70 percent of the patients treated with Hydrus were still medication-free four years after implantation. This means their glaucoma was controlled with the Hydrus alone and that the patients didn’t require topical glaucoma agents to maintain target IOPs.
Reasons to select MIGS
There are many reasons to select a MIGS procedure like the Hydrus for your glaucoma patients about to undergo cataract surgery. In addition to eliminating or reducing the need for topical medications, increasing adherence, and reducing the potential incidence of OSD, it may prevent more invasive incisional forms of glaucoma surgery in the future and may preserve more treatment options for the patient.
Rendia’s Exam Mode is a quick and easy way to show patients what glaucoma is and how it can be treated during cataract surgery—the one opportunity patients have to get MIGS.
Rendia is an essential part of increasing patient awareness, because providers can use Exam Mode’s visuals to demonstrate what glaucoma is and how it can be treated during cataract surgery. Exam Mode lets you fly through the simulated 3D anatomy model and animate functions and conditions to accompany your patient conversations. You can draw on the animations to explain concepts the way you want. It’s a quick and easy way to counsel patients on their options and show them how you can improve their quality of life.
The timing is critical. If you don’t counsel patients about MIGS at the time of cataract surgery, then you can’t do it.
Best candidates for MIGS
Candidates for MIGS include patients that are about to undergo cataract surgery with open angles (grade 3 or 4 on the Shaffer grading system) and have a clear media for adequate visualization. Patients that are contraindicated include eyes with traumatic, malignant, uveitic, or neovascular glaucoma or discernible congenital anomalies of the anterior chamber (AC) angle. The complication rates are minimal but any of the following should be monitored for: hypotony, hyphema, device malposition, chronic inflammation or pain and advancing glaucoma or uncontrolled IOP.
Candidates for MIGS include pre-surgery cataract patients with open angles. Complication rates are minimal, and the opportunities and benefits are significant.
These procedures have numerous advantages and need to be top of mind as patients only have one chance to receive this procedure. The opportunities and benefits are significant, the results are impressive, and the care of these patients is relatively straightforward. It’s a procedure worth considering for almost all of your cataract patients with primary open-angle glaucoma.
The views expressed above are of Dr. Paul Karpecki and do not necessarily reflect those of Rendia.
Your glaucoma patients may need a refresher on what the condition is and why it’s essential to treat it.
Educate them with this short video—watch it now.